16
Introduction and advantages
Eastman receives many questions about molding Eastman Tritan™ copolyester with hot runner systems. It can be a complex topic, loaded with variables that include not only mold design, but also part size, polymer formulation, and part design. Success depends on early collaboration and clear communication between Eastman, molders, tool builders, and hot runner suppliers.
We’re usingTritanMoldIt.com as a platform to jumpstart this communication between stakeholders. During the next few months, the Tritan experts will post a series of blogs, beginning with this overview and advantages of hot runners for amorphous copolyesters, including Tritan.
We invite you to help shape our series of hot runner blogs. Send your feedback. Ask your questions. Share your success stories. We look forward to hearing from you.
Key advantages of hot runner systems
In a hot runner molding system, the runners are within the mold. They are heated to maintain the plastic resin in a liquid state (a temperature above the material’s softening temperature, Tg) within the tool until it passes through the gate into the mold cavity. Unlike a cold runner system, hot runners are not ejected with the finished part. Instead, they stay in the mold in a liquid state, ready to fill the cavity on the next shot.
Hot runner systems are popular for high-production runs or in high-cavity molds, where quality is critical and gate witnesses are objectionable. A hot runner system can offer these advantages:
• Less resin used because there is virtually no waste
• Shorter flow path
• Reduced injection time; reduced open-dwelling time
• Lower cost per part for high-volume production—especially small parts
• Higher part quality
• More design flexibility
In Part 2 of our hot runner series, we’ll start focusing on specific ways for getting the best results with Tritan in a hot runner system.
| TMI TIP: Help make this ongoing series on hot runners more relevant for your needs. Let us know your feedback or questions—and share your experience with hot runner systems and Tritan. |
For more information about hot runners, download the Eastman Tritan™ copolyester Processing Guide.
9
If you have more questions about flowlines or other molding defects, talk with your Eastman technical service representative—and ask how to receive your free copy of our Injection Molding Troubleshooting Guide.
| TMI TIP: For more information about getting the best results when you mold with Tritan, watch the webinar Copolyester Processing Solutions. |
26
Consider these characteristics when selecting an adhesive:
- Chemical compatibility with parts being joined
- Aesthetics of the finished joint
- Expansion/contraction with temperature changes (NOTE: if expansion/contraction are a major concern, consider mechanical fasteners.)
- Brittleness, rigidity, flexibility
- Durability/service life
- Adhesive strength (adhesion to the plastic)
- Cohesive strength (resistance to internal tearing)
- End-use requirements
Eastman Tritan™ copolyester offers outstanding chemical resistance and has been studied with many adhesives and different bonding procedures.
Adhesive bonding procedures for Eastman Tritan™ copolyester
When adhesive bonding parts made with Tritan, joining surfaces should:
- Fit well without forcing
- Have no visible gaps
- Be smooth but not polished:
- Sand the surfaces you wish to bond with a 120-grit or finer paper.
- Diamond-wheel polishers, jointers/planers, or other mechanical devices can produce excellent results.
- Do not use soft-polishing wheels or flame polishing, as they can round the edges, creating gaps and improper fit.
Before selecting an adhesive system, conduct a careful evaluation of the part application to ensure the adhesive provides both acceptable performance (bond appearance and strength) and that it meets all applicable regulatory requirements. See inset box for several recommendations.
NOTE: This list of recommended adhesive systems does not include all products have been used successfully with Tritan. For best performance, always follow the guidelines provided by the adhesive manufacturer.
|
Recommended adhesive systems for use with Tritan
|
|---|
*Tested according to ASTM D1002
|
For details about bonding Tritan to itself and to other materials, contact your Eastman technical representative. For more information about adhesive bonding as well as tips for decorating, painting, overmolding, and other tips, download a free copy of the Secondary operations guide.
| TMI TIP: Adhesives that contain a volatile component may shrink while curing. To compensate for shrinkage, cut the joint on an angle, thereby providing space for the joint to be slightly overfilled. Consult your adhesive supplier literature for specific information on shrinkage. |
11
Electron beam (e-beam) radiation is one of the most popular methods for sterilizing medical devices—along with gamma radiation, ethylene oxide (EtO), autoclave, and low-temperature hydrogen peroxide gas plasma.
Eastman conducted studies to determine the effects of e-beam radiation on the optical and physical properties of Eastman Tritan™ copolyester and other transparent medical polymers.
| Formula | Resin type |
|---|---|
| Eastman Tritan™ copolyester MX711 | copolyester |
| Eastar™ polyester MN052 | polyester |
| Eastar™ copolyester MN211 | copolyester |
| Eastar™ copolyester MN006 | copolyester |
| DuraStar™ polymer MN611 | copolyester |
| Tenite™ propionate 360A40012 | Cellulose acetate propionate (CAP) |
| Lipid resistant PC | polycarbonate |
| TABS | Transparent ABS |
| Medical grade acrylic | Acrylic (PMMA) |
The objective of sterilizing medical devices is to reduce the bioburden to a safe level with minimal effects on the desired properties of its constituent polymers.
Technology advances have improved the performance of e-beam radiation, making it a safe, efficient, and reliable source of energy for sterilization. High dose rates reduce the time of exposure, which can make e-beam a less costly alternative to gamma radiation.
To study the effect of e-beam radiation on Tritan MX711, Eastman measured color shifting and physical property retention after e-beam radiation.
Optical properties
Samples were exposed to e-beam radiation (25 and 50 kGy), then stored in darkness and their color measured at Days 3, 7, 14, and 42 using HunterLab UltraScan™ Sphere 8000 and the CIE L*, a*, b* color scale. (Samples were stored in darkness and only exposed to light for color measurement.)
Figure 1 shows the changes in b* value, which measures the blue to yellow scale after e-beam exposure at 50 kGy. Photos of the molded samples before and after exposure are available upon request.
Figure 1. Eastman Tritan™ copolyester and other transparent resins after e-beam exposure at 50 kGY—b* color measurements
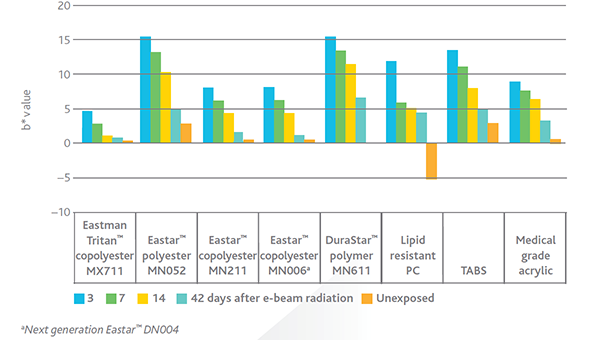
Figure 2 shows the difference in b* color values between unexposed samples and sterilized samples at Day 42—after e-beam radiation at both 25 and 50 kGy.
Figure 2. Change in b* color 42 days after e-beam radiation—25 and 50 kGy
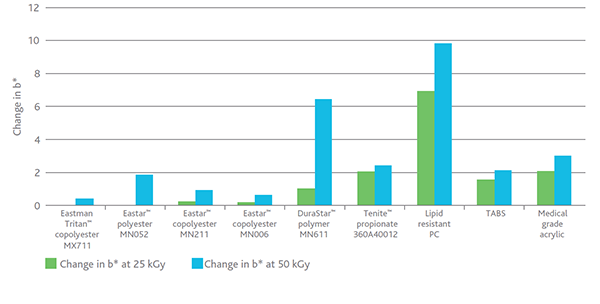
Physical properties were measured before and after e-beam sterilization at 25 and 50 kGy. No physical property degradation was noted for Tritan MX711 or the other resins tested. The polyesters and copolyesters in the study showed no statistical change in molecular weight although Tenite™ propionate 360 did show a loss of molecular weight, as expected from an aliphatic polymer.
For complete details of these studies or more information about the advantages of Eastman Tritan™ copolyesters, email Eastman today.
23
Use this handy mobile app to:
- Calculate drying time
- Calculate melt residence time
- Calculate shot capacity utilization
- Download data sheets
- Watch videos
- Get advice from the Tritan experts, and more.
Get your hands on the Tritan Mold It app.
This handy processing resource helps you get the best results from Eastman Tritan™ copolyester—and access valuable information on other engineering polymers.
The Tritan Mold It app’s Quick Links function also makes it easy to dive deeper into Tritan applications, successes, and the latest tips for molding Tritan.
Download the free Tritan Mold It app now.

| TMI TIP: After you’ve used the Tritan Mold It app, use the “App Feedback” feature to tell us how you like it—and provide any suggestions for making it an even more valuable tool for you. |




 Close
Close


