24

In case you missed this informative webinar on 11 May 2016, it is now available for you to watch whenever you choose. The webinar addresses in detail:
• The need for chemical resistance in connectors, stopcocks, and filtration components
• How engineering polymers compare for compatibility with drugs, lipids, and carrier solvents
• How to use Eastman Tritan™ copolyesters in your fluid management components to improve patient safety
In this one-hour webinar, Tritan experts explain the increasing pressure on today’s medical devices—especially fluid management components and blood contact devices—to meet high performance standards for:
• Patient safety
• Performance expectations (aesthetics/functionality)
• Portability and constant handling
• Aggressive disinfectants, harsh pharmaceuticals, and carrier solvents
• Sterilization (EtO, gamma irradiation)
• Changes in ISO 80369/AAMI CN3(PS):2014 standards for small bore connectors across all fluid management platforms
• Lifetime value
They also will discuss tests used to verify compatibility with disinfectants, harsh oncology drugs and their carrier solvents, and solvents used in fabrication and assembly.
Watch the webinar now!
| TIM TIP: For more information about chemical resistance in the medical market go to the Product Overview blogs and find “Why devices are failing in oncology drug delivery applications” or “Chemical Resistance—Parts 1 and 2”. |
5

The use of overmolded soft-touch materials is commonly employed to add both functional and decorative elements to articles produced from rigid thermoplastic elastomers (TPE). Eastman Tritan™ copolyester demonstrates excellent adhesion with commercially available TPE grades. Before selecting the TPE grade for use, work with either the TPE supplier or Eastman to choose a grade which is formulated for use with a copolyester substrate—and designing your part for successful results.
Part design considerations:
- Optimize part thickness and TPE thickness for adhesion and dimensional stability. TPE thickness in excess of the Eastman Tritan™ copolyester part thickness could result in warpage when you remove it from the mold. The typical rule of thumb recommends a substrate thickness twice that of the TPE.
- Mechanical interlocks can be incorporated to improve TPE adhesion and promote part durability. Mechanical interlocks become particularly important with thin TPE layers or very demanding fitness-for-use requirements.
- For designs incorporating soft-touch features on multiple surfaces, flow-through designs should be used to improve adhesion and durability.
- To minimize the potential for peeling or delamination, the edge of the TPE should be flush with or below the level of the non-overmolded section of the rigid substrate.
Be sure to consider the specific end-use environment of the overmolded article when selecting the appropriate TPE grade to confirm that it means your specific fitness-for-use criteria.
| TMI TIP: Stop by booth 2480 at the National Restaurant Association (NRA) show May 21-24, 2016. Tritan experts will be on hand to answer all your questions about Tritan. Then take your swings at the Eastman Batting Cage. |
For more information about using Tritan in secondary operations, as well as tips for decorating, painting, overmolding, and printing techniques, download a copy of our Secondary operations guide.
19
| TMI TIP: Proper cooling is well worth the wait when processing copolyesters. Their relatively low flexural modulus gives them a greater ability to flex and rebound—and allowing the skin to freeze is the best way to ensure optimum results. For more information, watch the webinar Copolyester Processing Solutions. |
If you have more questions about warped or distorted parts, talk with your Eastman technical service representative—and ask how to receive a free copy of our Injection Molding Troubleshooting Guide.
6
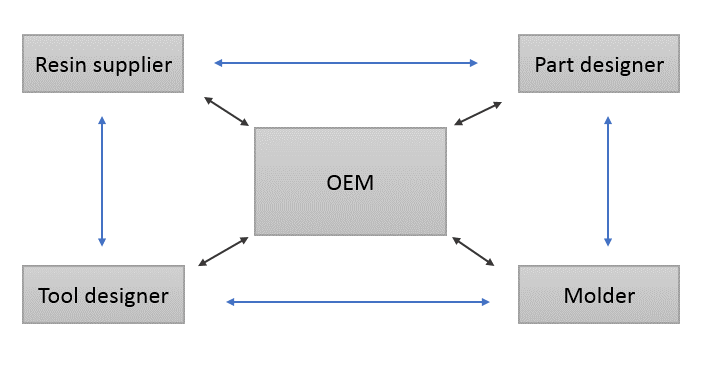
You can increase the chances of success in injection molding projects with early involvement of all major stakeholders. The many rewards of early collaboration include:
- Reduced product development time
- Optimized part performance
- Optimized cycle time
- Lower scrap rate
Eastman Design Services can add value starting today.
Eastman is a world leader in polymer innovation. We also have unmatched expertise in helping OEMs, part designers, tool designers, and molders receive the best processing efficiency and end-product quality.
Eastman Design Services can provide expertise in these areas:
- Part design reviews
- Resin selection assistance
- Mold filling simulation
- Stress analysis
- Injection mold tooling design reviews
- Secondary operations suggestions (mechanical fasteners, adhesives, ultrasonic welding, laser welding, spin welding, and more)
Early involvement of Eastman can help at critical steps in the project development flow—and help prevent false starts throughout the process.
If you have more questions about how early collaboration can help you, talk with your Eastman technical service representative.
| TMI TIP: Learn more about designing for manufacturability—a key step in project development. Watch the video of a recent webinar hosted by Eastman. |
28

Eastman will be presenting and exhibiting. Molding 2016 is the premier conference focused on innovations in injection molding technology—with information you can’t find anywhere else. That makes it the perfect place to show and tell the unique advantages of Eastman Tritan™ copolyester.
Join the Tritan experts Mar. 29 to 31, 2016, to learn how Tritan can improve your processes and end products.
Don’t miss our presentation.
On Thursday Mar. 31, Eastman representitives will show the advantages of Tritan in a first-of-its-kind mold for intricate medical parts. The presentation, titled “Validating the use of Eastman Tritan™ copolyester in high-precision, multicavity, hot runner medical molds,” will be held 8:30-9:30 AM. Whether you’re molding medical parts or you want efficient processing of innovative non-medical parts, you’ll find a lot of great information.
Don’t miss the chance to talk to a Tritan expert in person.
Eastman will be exhibiting throughout the three days of Molding 2016. Tritan experts will be available to answer questions about designing parts and molds to make the most of the advantages of Tritan.
| TMI TIP: When you stop by our exhibit at Molding 2016, be sure to ask about the Tritan Mold It mobile app. |




 Close
Close


